Aug 18, 2015
Introduction
 By Steven E. Finkelstein, MD
By Steven E. Finkelstein, MD
21st Century Oncology
 Sarah E. Hoffe, MD
Sarah E. Hoffe, MD
Moffitt Cancer Center
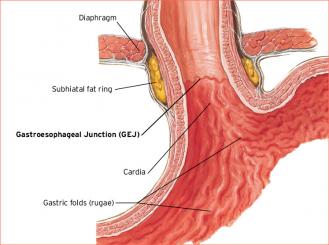
The optimal treatment of patients with clinically staged T2NO adenocarcinoma of the gastroesophageal junction (GEJ) is an elusive key question. This is, no doubt, partly due to the relative inaccuracy of endoscopic staging in patients with early-stage disease, coupled with a paucity of literature on the topic. The seminal problem of understaging patients who proceed to upfront resection is that it leads to consideration of adjuvant therapies that can involve larger radiotherapy volumes and concern for increased rates of toxicity. Similarly, overstaging may expose patients to unnecessary morbidity since their disease may have been eradicated with surgery alone.
In a contemporary era in which molecular patient selection for esophageal cancer treatment has not fully dawned, the treatment of choice for this patient population remains distinctly controversial. Whether clinical factors in addition to the endoscopic ultrasound stage will prove useful to guide the development of treatment algorithms for these patients is not currently defined.
In this issue of ASCO Connection, Dr. Manisha Palta, Dr. Brian G. Czito, and Dr. Christopher G. Willett discuss the neoadjuvant chemoradiation approach to clinically staged T2NO adenocarcinoma of the GEJ.
Dr. Finkelstein is a radiation oncologist and the Chief Science Officer and National Director of the Translational Research Consortium (TRC) at 21st Century Oncology in Arizona. He also serves as an Adjunct Associate Professor at Translational Genomic Research Institute (TGEN) and Editor-in-Chief of the journal Contemporary Radiation Oncology. He has been an ASCO member since 2003.
Dr. Hoffe is the Section Head of GI Radiation Oncology and an Associate Member of the faculty at Moffitt Cancer Center. She is also an Associate Professor in the Department of Oncologic Sciences at the University Of South Florida, Morsani College of Medicine.
Evidence for the Use of Neoadjuvant Chemoradiation and Surgery in the Treatment of T2N0 Gastroesophageal Junction Cancer



By Manisha Palta, MD, Brian G. Czito, MD, and Christopher G. Willett, MD, FASCO (left to right)
Duke University School of Medicine
Patients with T2N0 esophageal cancer comprise a rare subset of patients with localized esophageal cancer. This group illustrates the significant inaccuracies associated with preoperative clinical evaluation and high rates of pathologic upstaging in approximately one-half of patients at resection.
Although earlier randomized trials evaluating the role of neoadjuvant chemoradiation (CRT) and surgery for patients with esophageal and gastroesophageal (GE) carcinoma have yielded conflicting results, the largest study evaluating CRT—the Chemoradiotherapy for Oesophageal Cancer Followed by Surgery Study (CROSS)—clarified inconsistent results from prior trials.1-4
Three hundred sixty-eight patients with potentially resectable esophageal and GEJ adenocarcinoma and squamous cell carcinoma (SCC) were randomly assigned to CRT with carboplatin/ paclitaxel preoperatively or to surgery alone. Patients with clinical stage T1N1M0 or T2-3N0-1M0 were enrolled. Participants’ disease was rigorously staged using pulmonary function tests, upper endoscopy, endoscopic ultrasound, CT of the neck, chest, and upper abdomen, and neck ultrasound. Radiotherapy (RT) was delivered to a total dose of 41.4 Gy in 23 fractions.
The primary endpoint, overall survival (OS), was significantly longer in the CRT arm: median OS was 49 months in the CRT arm compared with 24 months in the surgery-only cohort. Trial subgroup analysis indicated a greater benefit of neoadjuvant CRT in patients with clinical N0 disease. Overall, the pathologic complete response (pCR) rate was 29%, with higher R0 rates seen in the CRT arm, without additional postoperative morbidity. This regimen was well tolerated, with over 90% of enrolled patients completing the intended neoadjuvant treatment course.5
Patterns of treatment failure from the CROSS study demonstrated the rate of overall locoregional recurrence after CRT and surgical resection to be 14%, whereas the isolated, in-field recurrence rate was 1%.6 This landmark trial established the efficacy of neoadjuvant CRT and surgery in the treatment of esophageal and GEJ cancer, including for patients with T2N0 cancer.
Although there is a benefit to neoadjuvant CRT prior to surgical resection, the efficacy of alternative neoadjuvant strategies is less well defined. To date, only one phase III randomized trial— the Preoperative Chemotherapy or Radiochemotherapy in Esophagogastric Adenocarcinoma Trial (POET)—has evaluated neoadjuvant chemotherapy alone with CRT. In this German study, 126 patients with locally advanced esophageal, GEJ, or gastric cardia adenocarcinoma were randomly assigned to chemotherapy followed by surgery versus chemotherapy, then CRT, followed by surgery. The study intended to enroll 354 patients but closed early secondary to poor accrual.
The upfront chemotherapy regimen consisted of combination 5-FU/CDDP/ leucovorin (5-fluorouracil/cisplatin/leucovorin). The CRT regimen consisted of etoposide and CDDP administered concurrent with RT to a limited dose of only 30 Gy.
As expected, the pCR rate was higher in the trimodality arm compared with preoperative chemotherapy alone (16% versus 2%). Additionally, there were significantly higher rates of R0 resections and fewer patients with node-positive disease in the CRT cohort. The study’s primary endpoint was OS and, given limited accrual, was underpowered to detect a difference. Despite this, the three-year OS was 47% in the CRT/surgery arm compared with 28% in the chemotherapy arm— trending to a clinically significant difference (p = 0.07).7
Similarly, an updated meta-analysis of more than 4,000 patients accrued to 24 randomized trials evaluated the role of neoadjuvant CRT versus neoadjuvant chemotherapy alone in patients with resectable esophageal and GEJ cancer. An all-cause mortality benefit of 8.7% at two years was seen in patients receiving neoadjuvant CRT, with survival benefits similar between patients with either squamous or adenocarcinoma histologies. By comparison, in patients receiving neoadjuvant chemotherapy alone, estimated absolute survival difference at two years was 5.1%, which was only significant for patients with adenocarcinoma.8
Advocates of neoadjuvant chemotherapy alone may cite the recently published French Francophone de Cancérologie Digestive (FFCD) 9901 trial that evaluated the role of neoadjuvant chemoradiation in patients with clinically staged early esophageal cancer. This study compared surgery alone or neoadjuvant CRT (45 Gy with concurrent cisplatin and 5-FU at weeks one and five in 195 patients with stage I/II, predominantly SCC esophageal cancer).
On interim analysis the trial was closed for futility, and at median follow-up of seven years, there were no significant differences in three-year survival or R0 resection rate.9 However, this trial was underpowered for the primary endpoint of OS, and the excess postoperative mortality seen in the FFCD trial is much higher than reported in the CROSS study, likely influencing study outcomes in this group.10
Results from the CROSS study, the POET study, and large meta-analyses strongly support the use of neoadjuvant CRT. Randomized data evaluating alternative strategies such as neoadjuvant chemotherapy are limited but do not suggest a clinically significant improvement (or arguably, even equivalence) in survival compared with CRT. Ongoing studies of neoadjuvant chemotherapy and CRT may further clarify the optimal neoadjuvant therapy. In 2015, however, there is compelling evidence regarding the value of neoadjuvant CRT and surgery in the treatment of T2N0 esophageal and gastroesophageal cancer.
Dr. Palta is an Assistant Professor of Radiation Oncology at Duke University School of Medicine.
Dr. Czito is the Gary Hock and Lyn Proctor Associate Professor of Radiation Oncology at Duke University School of Medicine. An ASCO member since 2002, he was Chair of the Esophagus and Stomach Track of the 2013 Gastrointestinal Cancers Symposium and a member of the 2012-2014 Gastrointestinal Cancers Symposium Program Planning Committee.
Dr. Willett is the Chair of the Department of Radiation Oncology at Duke University School of Medicine. An ASCO member since 1988, he is the Past Chair of the Gastrointestinal Cancers Symposium Steering Committee and has served as a member of multiple committees, including the Gastrointestinal Cancers Symposium News Planning Team Committee and the Cancer Education Committee. He received the distinction of Fellow of the American Society of Clinical Oncology (FASCO) in 2011.
References
1. Walsh TN, Noonan N, Hollywood D, et al. N Engl J Med. 1996;335:462-7.
2. Urba SG, Orringer MB, Turrisi A, et al. J Clin Oncol. 2001;19:305-13.
3. Tepper J, Krasna MJ, Niedzwiecki D, et al. J Clin Oncol. 2008;26:1086-92.
4. Burmeister BH, Smithers BM, Gebski V, et al. Lancet Oncol. 2005;6:659-68.
5. van Hagen P, Hulshof MC, van Lanschot JJ, et al. N Engl J Med. 2012;366:2074-84.
6. Oppedijk V, van der Gaast A, van Lanschot JJ, et al. J Clin Oncol. 2014;32:385-91.
7. Stahl M, Walz MK, Stuschke M, et al. J Clin Oncol. 2009;27:851-56.
8. Sjoquist KM, Burmeister BH, Smithers BM, et al. Lancet Oncol. 2011;12:681-92.
9. Mariette C, Dahan L, Mornex F, et al. J Clin Oncol. 2014;10:2416-22.
10. Czito BG, Palta M, Willett CG. J Clin Oncol. 2014;32:2398-2400.